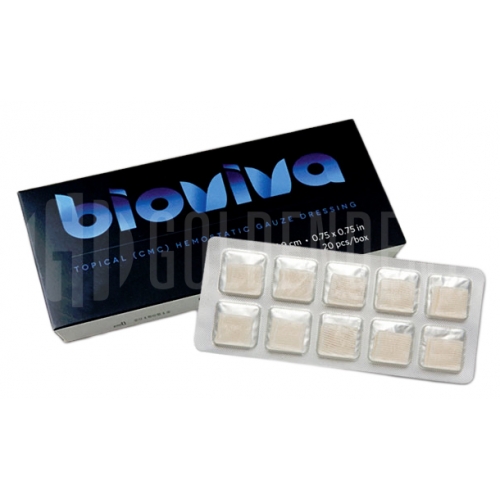
Bioviva Hemostatic Gauze Wound Dressing


Hemostatic Wound Dressing to Promote Optimal Healing
Surgical dressings and hemostatic materials are available in a wide range of compositions and brands including plugs, powders, tapes, strips or sponges. All of these products are coated and/or treated with various hemostatic materials where the intent of such products is to control bleeding and to promote efficient healing with our patients following an extraction. While some of these products are resorbable and will safely resorb on their own, non-resorbable materials are also available and would need to be removed following healing. As with any dental products, it is important to note which type of material you are using and to know the specifications and uses of each product as they are not all the same. Over the years I have found that the product marketing is similar for such products, but the clinical results, ease of use and product costs are not the same. As dentists we need to be aware of this. I have found that the Bioviva Hemostatic Gauze Wound Dressing is the optimal choice from clinical results, ease of use and cost effectiveness perspective for certain cases within my practice.
Bioviva is individually packaged, sterile, soluble, cellulose based, bacteriostatic hemostatic gauze dressing made from oxidized, regenerated cellulose derived from plants. Bioviva absorbs blood and immediately transforms into a viscous gel, sealing the extraction site, filling wound voids, seals capillary ends and activating the clotting system to assist the body to stop bleeding (Figure 1). As noted above, the main factors I look to in evaluating a product are clinical results, ease of use and lastly the cost effectiveness of such product. Of course a better priced product is ideal for the practitioner's bottom line, but with many products you often get what you pay for and the clinical results and patient aspects should always be considered first.
Bioviva Clinical Results
If you have ever heard my lectures, webinars, read my past clinical articles, or watched some of my videos, you have likely heard me be an advocate for placing some sort of a grafting material in a socket following an atraumatic extraction. With today's materials and training available to us, I strongly recommend that you offer grafting in your office. However, I acknowledge that for various reasons including the type of procedure (e.g., third-molar extractions, pediatric extractions), the patient's financial resources, or the practitioners' inexperience with grafting that not all extraction sites will be grafed by many practitioners. This is where a product like Bioviva can easy be implemented to prevent dry-socket or any other post-operative complications, and most importantly to minimize patient discomfort. Bioviva is efficient, effective and safe to use. In my experience, it coagulates blood faster than other hemostats I have used and is very effective to stop bleeding even when the patients may be taking anticoagulants. It allows for reduced infection as it provides a barrier over the extraction site and it has been proven to nearly eliminate dry sockets. It is bacteriostatic and is very effective.
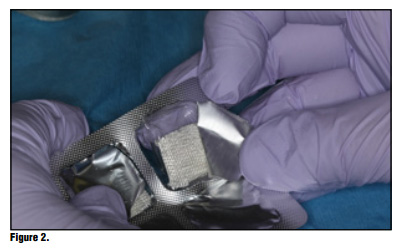
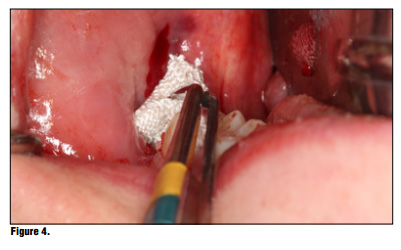
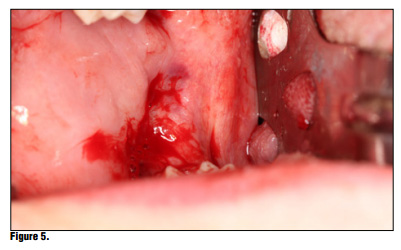
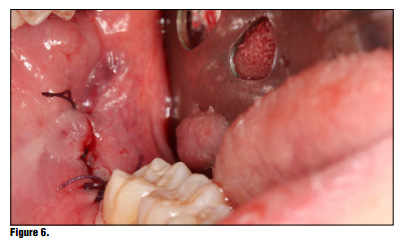
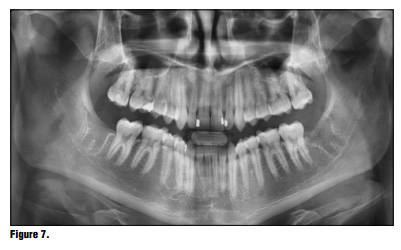
Bioviva Ease of Use Te product is packed in individual blister packs in 1.9 cm squares for ease of use and to avoid any cross contamination issues (Figure 2). Simply puncture the packaging backing and remove the gauze from the packaging using dry forceps to avoid the gauze from sticking to the forceps (Figure 3). The product can be cut to size if the gauze is larger than the site, but I have found this is generally not necessary and the product can be folded to properly ft using tissue forceps (Figure 4). Once the product is removed from the packaging the product is simply placed onto the wound or extraction site dry and with a matter of 15-20 seconds the material absorbs the wound fluids and turns into a gel and fills the void (Figure 5). Once the site is properly sealed with the material it can be properly rinsed and should not become dislodged. In this case, a suture was placed in conjunction with the product, but in many cases this may not be necessary. A practitioner's judgment should be utilized in determining if a suture is required in conjunction with the product (Figure 6). Te site can be sutured, but is not necessary, and it should not be sutured in place using primary closure as the product needs to be in contact with oral environment and have proper drainage. It is not recommend to suture with primary closure as the creation of a closed contaminated space could be created and this may increase the risk for abscess formation. Digital panoramic x-ray shows post extraction view of the patient's third-molar extraction sites (Figure 7).
Bioviva Cost Effectiveness
As previously noted, the clinical results are the number one factor for evaluating a product, but as a practice owner we must also consider our costs. Bioviva is priced much better than other products on the market - but more importantly it provides great clinical results. With Bioviva it can also eliminate or reduce patient follow-up visits for discomfort issues resulting in less chair time as well as post-operative visits for suture removal if a suture is not utilized with Bioviva based on the case.
Overall, I recommend learning more about Bioviva and its benefits by visiting www.goldendentalsolutions.com or calling 877- 987-2284 to discuss with one of their product specialists. It has been a great product addition to my practice for certain cases.

Bioviva Hemostatic Gauze Wound Dressing
Prices change with available options